Only Authentic Diamonds
The Nature of the Diamond
Natural diamond crystals formed millions of years ago inside the Earth, at about 160 km deep, and were brought to the surface long after through volcanic eruptions. These eruptions created narrow vertical channels of an igneous rock known as kimberlite.
Kimberlite pipes are excavated to extract the diamonds, and the extracted ore is subsequently mechanically broken down to release the crystals. The concentration of diamonds in kimberlite is very low, about one part per million, therefore, miners must extract large quantities of ore to obtain diamonds.
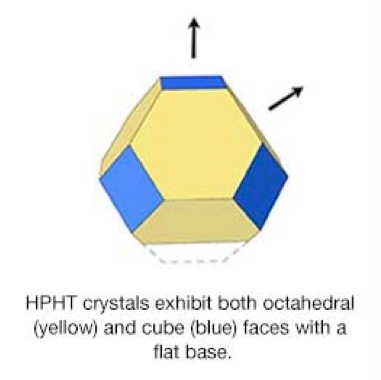
Natural diamonds form under specific conditions of pressure and temperature, the latter significantly higher than that used for growing synthetic diamonds. Thus, while at high temperatures natural diamonds develop into octahedral crystals, synthetic diamonds produced at lower temperatures tend to grow as crystals with both octahedral and cubic faces.
Synthetic diamonds are grown within a considerably short time frame, ranging from a few weeks to just over a month, in significantly different conditions from those of natural diamond formation in the depths of the earth. The short growth period results in the shape of synthetic diamond crystals being quite different from that of a natural diamond.
Synthetic diamonds
In the mid-1950s, the first scientists developed the foundations for the cultivation of synthetic diamonds, initially obtaining only small crystals. The production of crystals of larger dimensions, suitable for use in jewelry, began in the mid-1990s and continues to evolve.
Synthetic diamonds, cultivated in various countries around the world, find their main use not only in jewelry but also in industrial applications. The traditional synthesis method, known as high-pressure high-temperature (HPHT) growth, produces diamonds from a melted metal alloy, such as iron (Fe), nickel (Ni), or cobalt (Co).
The most recent method, called Chemical Vapor Deposition (CVD) or low-pressure high-temperature (LPHT) growth, involves the formation of diamonds from a gas inside a vacuum chamber.
In this environment, particles react with each other, depositing in layers of carbon that gradually consolidate into a single stone. In both methods, a diamond crystal or plate is used as a "seed" to initiate growth.

The growth of HPHT diamond occurs in a small capsule within a device capable of generating very high pressures. Inside the capsule, the starting material, diamond powder, dissolves in the flow of molten metal and crystallizes onto the seed to form the synthetic diamond crystal. The crystallization of one or a few crystals occurs within the timeframe of a few weeks to just over a month.
Synthetic HPHT diamonds typically display both cubic and octahedral faces. Since the shapes and internal growth patterns of natural and HPHT synthetic diamonds differ, the latter can be identified through these growth patterns, which represent one of the most reliable methods for distinguishing them.
Resulting faceted synthetic gems often exhibit distinctive visual features, such as color variations, fluorescence zonation, and "graining" patterns related to their cross-sectional growth structure, as well as the possible presence of dark metallic inclusions.
In some cases, the material exhibits phosphorescence that persists after the ultraviolet lamp is turned off. These synthetic diamonds can be reliably identified through laboratory techniques such as visible spectroscopy and photoluminescence.
Most crystals grown with HPHT are yellow, orange-yellow, or brownish-yellow. Nearly all are of type IIb, which is rare in natural diamonds. Creating colorless HPHT synthetics has been a challenge as modifications to growth conditions and equipment are required to exclude nitrogen.
Additionally, the growth rates for high-purity colorless diamonds (type IIa or weak type IIb) are lower compared to those of type Ib synthetic diamond, requiring longer growth times and greater control over temperature and pressure conditions. Although in the past it was difficult to grow high-quality colorless HPHT crystals, recent developments have produced crystals sufficient for faceted stones exceeding 10 carats in weight.
The addition of boron in the diamond growth system leads to the formation of blue-colored crystals. Other colors such as pink and red can be obtained through post-growth treatment processes, which involve exposure to radiation and subsequent heating, but such colorations are less common.
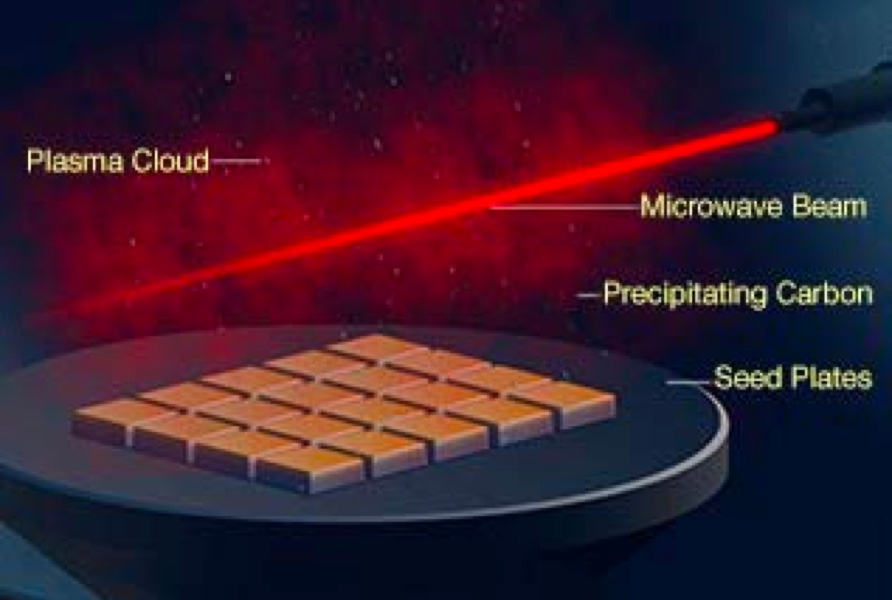
CVD diamond growth occurs inside a vacuum chamber filled with a carbon-containing gas, such as methane. A source of energy, such as a microwave beam, breaks down the gas molecules and carbon atoms are drawn downward by the flat diamond plates. Crystallization occurs over a period of several weeks to create a number of crystals; the exact number depends on the size of the chamber and the number of seeds.
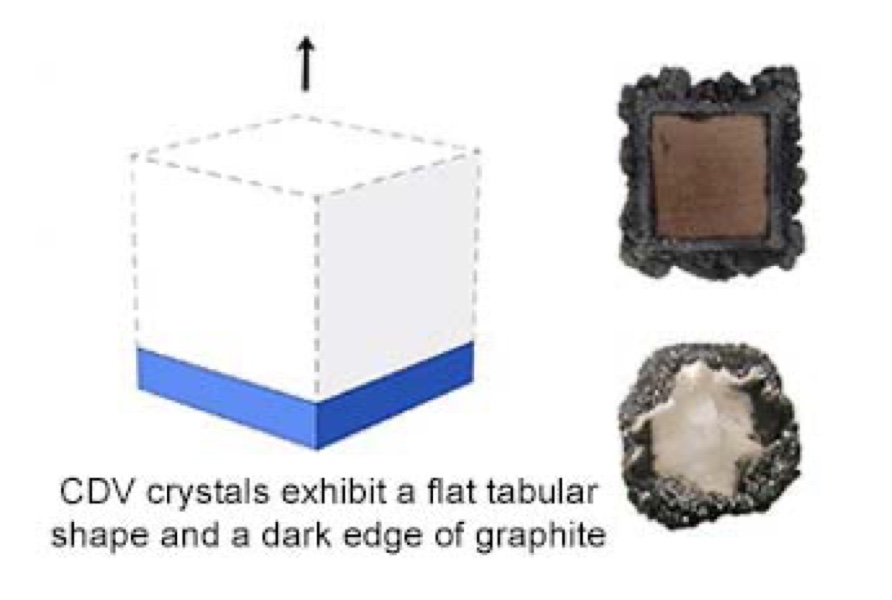
These tabular-shaped crystals often have a rough edge of black graphite. It is also common for them to have a brown coloration, which can be removed with heat treatment before faceting. Similar to HPHT synthesis, the CVD method is also evolving, allowing manufacturers to produce larger diamonds and improvements in color and purity.
In recent years, several companies have started producing synthetic diamonds for jewelry use. There have been improvements in clarity and color of these diamonds, as well as an increase in their carat weight. For the identification of gem materials of any type, an experienced gemologist employs various tools for gem testing.
These include a refractometer, a ultraviolet fluorescence lamp, a binocular microscope, a polariscope, and other testing instruments. With the constant improvement in the quality of synthetic diamonds, it becomes progressively more difficult to distinguish them from natural gems using standard equipment.
Although an experienced gemologist may find it difficult to recognize synthetic diamonds with the naked eye alone, there are several clues that may suggest a synthesis process. However, these require more in-depth analysis and often the use of specialized instrumentation.
These visual characteristics are typical of most synthetic diamonds, although not all faceted synthetic diamonds exhibit all of these peculiarities. For example, a particular synthetic diamond may not exhibit fluorescence. Therefore, it is crucial to base the identification of a synthetic diamond on as wide a range as possible of diagnostic characteristics.
Colored synthetic diamonds grown with HPHT often exhibit irregular coloration. This aspect can be observed using transmitted light in a microscope and, if necessary, immersing the cut stone in water or mineral oil to minimize surface reflections. Such color zoning is generally attributable to how impurities, such as nitrogen, are incorporated during formation.
Occasionally, natural diamonds show color zoning, but not in the geometric pattern exhibited by HPHT synthetic diamonds. Conversely, synthetic diamonds obtained with CVD typically show uniform coloration. HPHT synthetic diamonds often exhibit inclusions of solid melted metal, which appear black and opaque in transmitted light but have a metallic luster in reflected light.
The flux alloy used in HPHT diamond growth typically contains elements such as iron, nickel, and cobalt. Therefore, synthetic diamonds with larger metal inclusions may be attracted by a magnet. Synthetic diamonds grown using the CVD method, on the other hand, form differently and do not exhibit metal inclusions.
| Synthetic HPHT | Synthetic CVD |
|---|---|
| Distribution of non-uniform colors | Distribution of non-uniform colors |
| Different graining patterns | No graining pattern |
| Unusual fluorescence colors | Unusual fluorescence colors |
| Fluorescence color patterns | Fluorescence color patterns |
| Occasional phosphorescence | Occasional phosphorescence |
| Flux metallic inclusions | Occasional dark spots |
| No deformation pattern | Banding strain patterns |
| Possible inscription on girdle | Possible inscription on girdle |